
~ ~ where H V and hL are enthalpies of the pure component. For ideal solutions the enthalpies can be determined from the sum of the pure component enthalpies multiplied by the corresponding mole fractions: ~ (2-47a) HV ! yi H V Tdrum, pdrum The enthalpy hL and H V can now be calculated. L and V are determined from the overall massīalances. Once V F has been found, xi and yi 38) are calculated from (2-Īnd (2-39). If it is close enough to zero, the calculation is finished, otherwise, repeat the Newton convergence for the next trial. Once F k 1 is calculated the value of the Rachford-Rice V equation can be determined. Substituting the Rachford-Rice equation and (2-45) into (2-44) C K i 1zi 1 K 1V F V V i !1 i ! C K i 12 zi F k 1 F k ?1 K 1V F A2 i !1 i Then the derivative is df C K i 12 zi k ! (2-45) 2d F Vi !1 To use it, however, we need equations for both the function and derivative. This equation gives us the best next guess for the fraction vaporized. We desire to have f k 1 set ! 0 f k 1 equal to zero, so we
#DEPRIESTER CHART CALCULATOR FOR N PENTANE TRIAL#
Value, the equation for the Newton convergence isĭf k ( F f k 1 f k ! V d F Vwhere f k is the value of the function for trial k and df k d F is the value of the derivative of the function V for trial k. The Newton convergence procedure will converge faster. For V F instance, the Secant method can be used by selecting two values of and calculating the values of the V F summation (it will be zero only at the correct value of ). This equation can V F be solved by many different convergence procedures. V C K i 1zi f ! !0 F i !1 1 K 1V i FThis equation, which is known as the Rachford-Rice equation, has excellence convergence properties.Since the feed composition,zi are specified and i can K Tdrum be calculated when and are given, the only pdrum variable is the fraction vaporize. Subtracting the sum term by term, we have

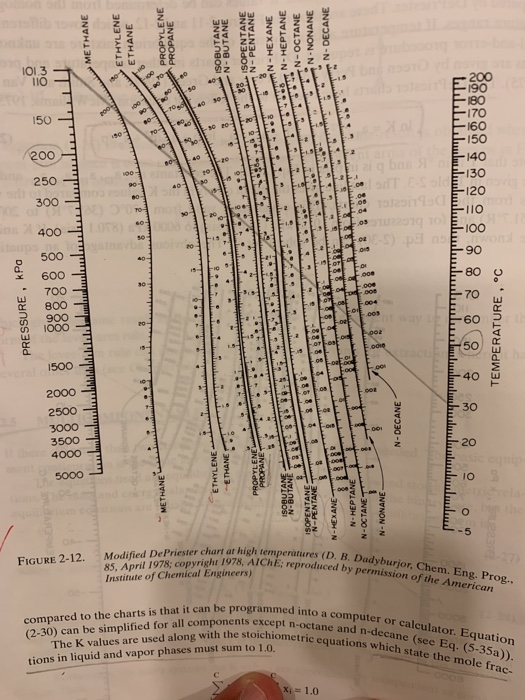
That is, ifV F wrong the is chosen, the F that is chosen next may not be V better. Although V the equations * and ** are both valid, they do not have the good convergence properties. If C is greater than 3, a trial-and-error procedure or root finding technique must be used to find F. To simplify the solution procedure, we first use yi y ! K i xi equilibrium, to remove Now there are only 22 equations to solve simultaneously. Since Tdrum and pdrum are known, the 10 Ki can be determined easily (say, from the DePriester charts or using the McWilliams relation). How does one solve 32 simultaneous equations? However we will restrict ourselves to ideal solution where Then we must find 10 K s, 10 x s, 10 y s one L and one V or 32 yi ! If pdrum and Tdrum or one liquid or vapor composition are also specified, then a sequential procedure can be used. Usually the feed, F, and the feed mole fractions z1 for C-1 of the components will be specified. Multicomponent Flash DistillationThe mass balances, The K values are used along with the stoichiometric equations which the mole fraction in liquid and vapor phases must equal to 1.0 33 for constantsMcWilliams, M.L., An Equation to Relate K-factors to Pressure and Temperature, Chem. The constants are given in the following table. The DePriester charts have been fit to the following equation (McWilliams, 1973):Ī p 2 a p3 aT 1 aT 2 ln K ! 2 aT 3 aP1 ln p 2 T T p pNote that T is in oR and p is in psia. thus,įor light hydrocarbons, the approximate K values can be determined from the monographs prepared by DePriester i i i where in generalġ These two equations are written once for each componentįortunately, for many systems the K values are approximately independent of composition. The mass and energy balances are very similar to those used in the binary case, but the equilibrium equations are usually written of K values. The equation used are equilibrium, mass and energy balances, and stoichiometric relations. Multicomponent Flash DistillationIf there are more than two components, an analytical procedure is needed.

